In this article, You will read Diamond and Graphite Distribution in India – for UPSC IAS.
Diamond
- Diamond has been the most valuable among gems for more than 2,000 years. Diamond is the hardest naturally occurring substance found on Earth.
- Diamond occurs in two types of deposits primarily in igneous rocks of basic or ultrabasic composition and in alluvial deposits derived from the primary sources.
- Diamonds are formed in the mantle. They brought to the earth’s crust due to volcanism. Most of the diamonds occur in dykes, sill, etc.
- Its composition is pure carbon and has a cubic crystal system and common form octahedron.
- India is known for its diamond cutting & polishing business, especially for small-sized diamonds. Most of the world’s diamond cutting and polishing business comes to India, particularly Surat in Gujarat. Indian Diamond Industry handles about 80% of the global polished diamond market.
- Being the hardest natural substance known, the industrial variety of diamonds are used in ornaments, grinding, drilling, cutting, and polishing tools.
- In addition, diamond exhibits the highest thermal conductivity amongst minerals and has high electrical resistivity making it suitable for application in semiconductors.
Diamond Distribution in India
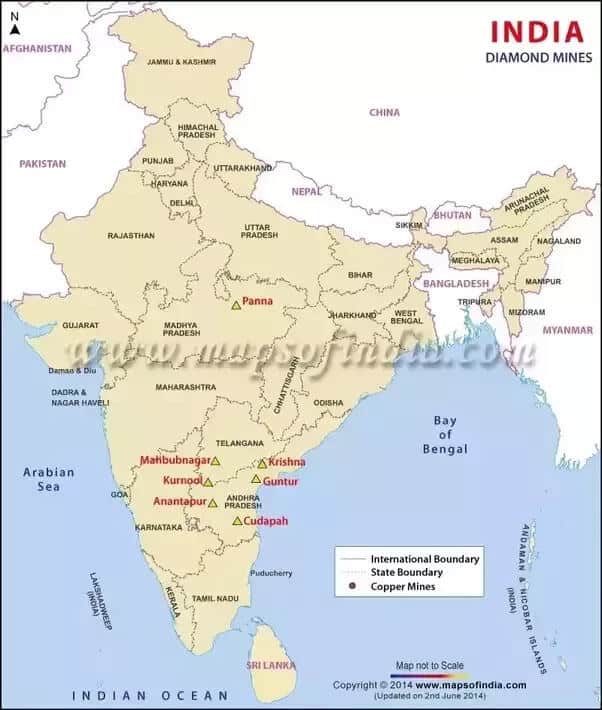
Diamond occurrences are reported since prehistoric times in the country. Presently, the diamond fields of India are grouped into four regions:
- South Indian tract of Andhra Pradesh, comprising parts of Anantapur, Kadapa, Guntur, Krishna, Mahabubnagar, and Kurnool districts;
- Central Indian tract of Madhya Pradesh, comprising Panna belt;
- Behradin-Kodawali area in Raipur district and Tokapal, Dugapal, etc. areas in Bastar district of Chhattisgarh; and
- Eastern Indian tract mostly of Odisha, lying between Mahanadi and Godavari valleys.
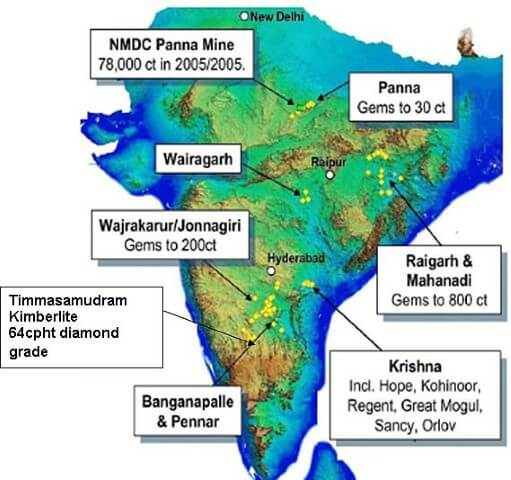
- Reserves have been estimated only in the Panna belt and Krishna Gravels in Andhra Pradesh.
- The new kimberlite fields are discovered recently in the Raichur-Gulbarga districts of Karnataka.
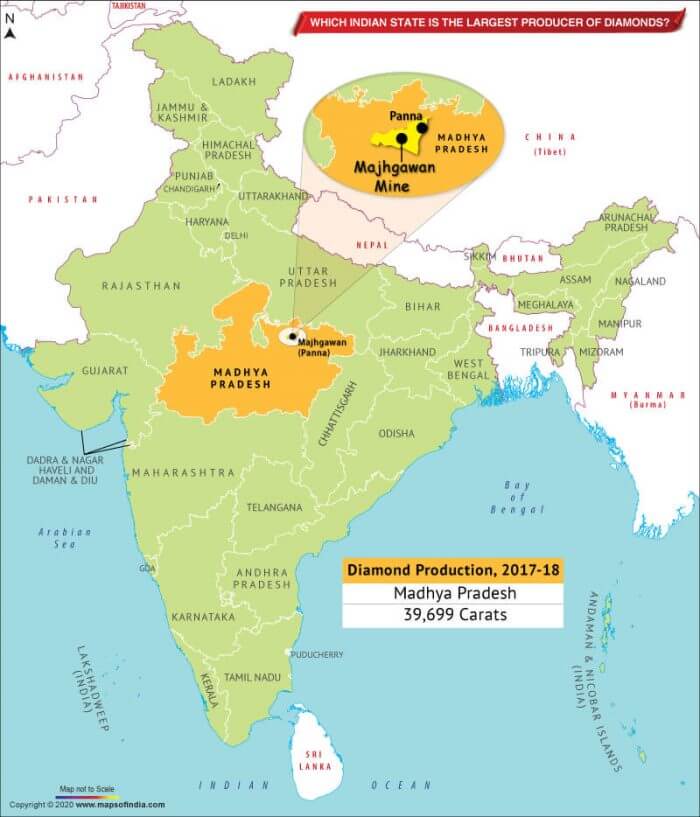
- Madhya Pradesh accounts for about 90.18% of resources followed by Andhra Pradesh 5.72% and Chhattisgarh 4.09%.
- In India, there is only one mine at Majhgaon in Panna (Madhya Pradesh) of NMDC for a production capacity of 84,000 carats and the total diamonds recovered from this mine so far are little more than 1 million carats.
Graphite
- Graphite is a naturally occurring form of crystalline carbon.
- Graphite is also known as plumbago or black lead or mineral carbon is a stable form of naturally occurring carbon.
- The carbon content in Graphite is never less than 95%.
- Graphite may be considered the highest grade of coal, just above anthracite.
- It is not normally used as fuel because it is difficult to ignite.
- It is found in metamorphic and igneous rocks.
- Graphite is extremely soft, cleaves with very light pressure.
- It is extremely resistant to heat and is highly unreactive.
- Most of the graphite is formed at convergent plate boundaries where organic-rich shales and limestones were subjected to metamorphism due to heat and pressure.
- Metamorphism produces marble, schist, and gneiss that contain tiny crystals and flakes of graphite.
- Some graphite forms from the metamorphism of coal seams. This graphite is known as “amorphous graphite”.
- Graphite is a non-metal and it is the only non-metal that can conduct electricity.
Applications of Graphite
- Natural graphite is mostly consumed for refractories, batteries, steelmaking, expanded graphite, lubricants, etc.
- A refractory material is one that retains its strength at high temperatures.
- Natural and synthetic graphite are used to construct the anode of all major battery technologies.
- The lithium-ion battery utilizes roughly twice the amount of graphite than lithium carbonate.
- Natural graphite in this end-use mostly goes into carbon raising in molten steel [to make steel stronger]
- Natural amorphous graphite are used in brake linings for heavier vehicles and became important with the need to substitute for asbestos.
- Graphite lubricants are specialty items for use at very high or very low temperatures.
- Modern pencil lead is most commonly a mix of powdered graphite and clay.
Indian Graphite Resources
India’s Graphite occurrences are found in states like Jammu and Kashmir, Gujarat, Jharkhand, Arunachal Pradesh, Karnataka, Kerala, Maharashtra, Tamil Nadu, Odisha, Chattisgarh, and Rajasthan.
As per GSI’s 2013 report,
- Arunachal Pradesh (43%),
- Jammu & Kashmir (37%),
- Jharkhand (6%),
- Tamil Nadu (5%) and
- Odisha (3%)
Operational Indian Graphite Resources
- Most of the Graphite Production is concentrated in these states
- Tamil Nadu (37%),
- Jharkhand (30%), [Palamu district in Jharkhand is the most important]
- Odisha (29%).
Active mining centres of graphite are in
- Jharkhand – Latehar & Palamu districts
- Odisha – Bargarh, Nuapada, Rayagada & Balangir districts
- Tamil Nadu – Madurai & Sivagangai districts
Differences Between Graphite and Diamond
| Diamond | Graphite |
| In diamonds, strong three-dimensional networks are formed due to the presence of covalent bonds. | Graphites are formed due to the weak van der Waals force of attraction. |
| Hard in nature | Soft in nature. |
| Since molecules are closely packed they have high density. | Because of the large gap between the molecules, they have low density. |
| Since there is no free carbon atom, the diamond does not conduct electricity. | Because of the presence of free carbon atoms in graphite, they can conduct electricity. |
| Diamond is 100% carbon. | Graphite contains 95% or more carbon. |
| Diamond (one of the most stable) is less stable than graphite. | Graphite is one of the most stable substances on earth. |

Very Nice compilation
So kind of you
ThAnk you